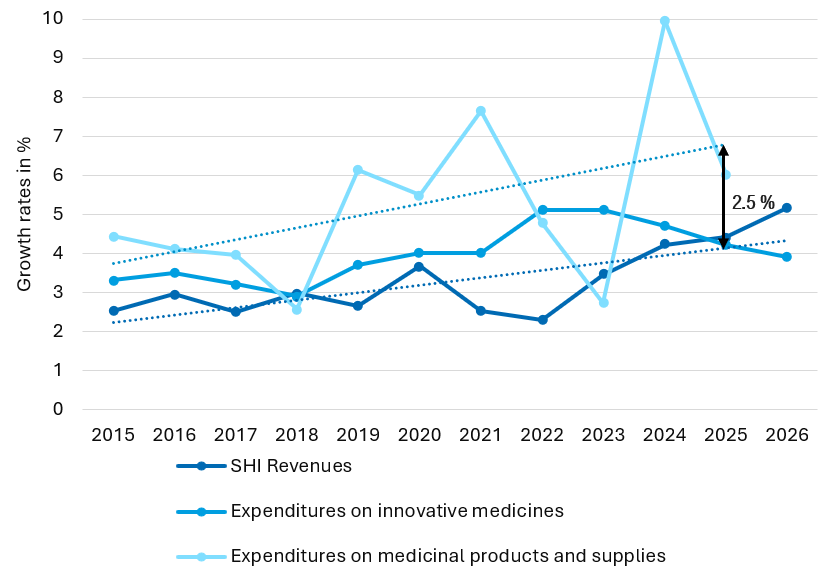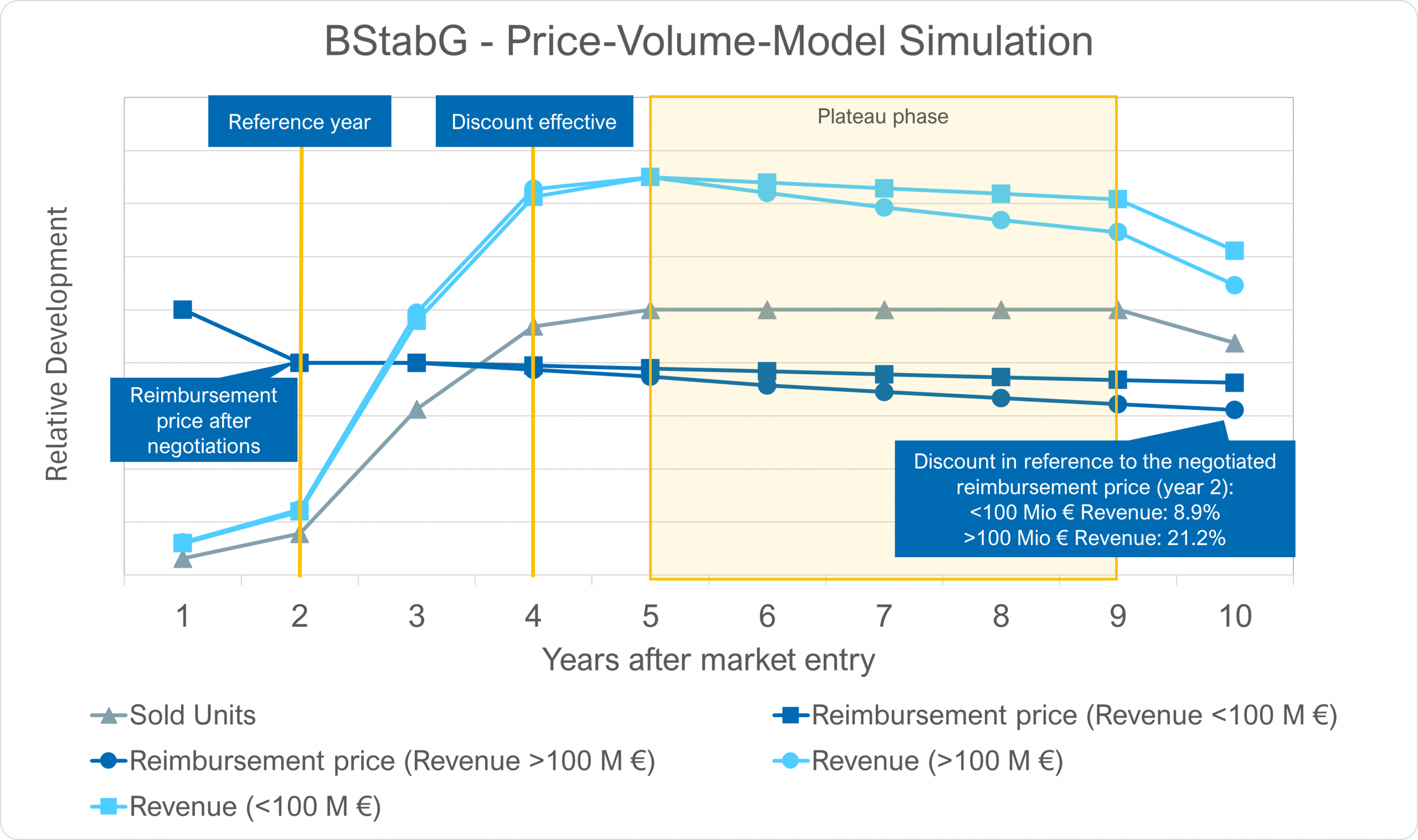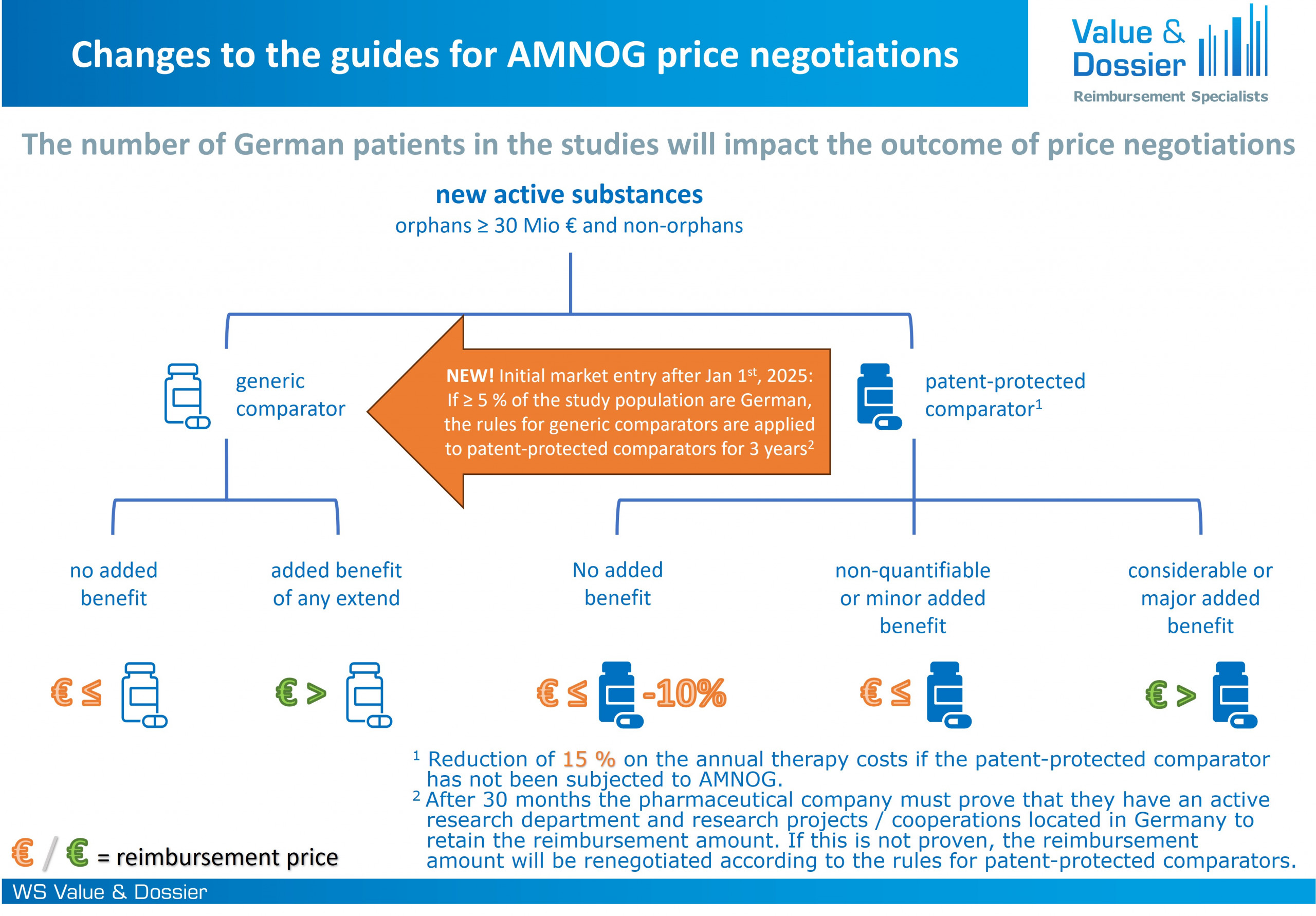
Figure 1: Different price guidelines apply for the AMNOG price negotiations if ≥ 5 % of the study population are German
In November 2022, the act on the financial stabilization of the statutory health insurance (GKV-FinStG) came into force, in which, among other things, the so-called AMNOG guidelines for negotiations on the reimbursement amounts of pharmaceuticals were introduced. These regulations tightened the legal framework for negotiations for pharmaceutical companies. The Medical Research Act (MFG) now exempts medicinal products for which ≥ 5 % German patients were included in the clinical trials relevant to the assessment from individual rules of the AMNOG guidelines. The potentially higher agreed reimbursement amount based on the adjusted rules is valid for 3 years. If a pharmaceutical company conducts active research in Germany, the agreed reimbursement amount can be maintained even after these 3 years. The requirements of active R&D aim to strengthen Germany as a research location. The first condition is that the pharmaceutical company has a permanent and active research department in Germany. In the case of development cooperations, licensing, or asset deals, it is also acceptable if the research department belongs to the respective cooperation or licensing partner. In addition to that, the second condition requires the pharmaceutical company to maintain relevant own projects (e. g. sponsored studies) and/or cooperations with public research institutions in Germany. Projects and/or cooperations must be ongoing; completed projects and/or cooperations will not be accepted. Pharmaceutical companies that do not have a research activity in Germany are obliged to renegotiate the reimbursement amount according to the strict AMNOG guidelines for patent-protected comparators at the end of the 3-year period.
In summary, medicinal products are exempted from the strict price negotiation guidelines introduced by the GKV-FinStG for a duration of 3 years if the studies used for the AMNOG assessment included ≥ 5 % German patients. This benefit can be maintained even after these 3 years if the pharmaceutical company actively participates in R&D in Germany. It seems unlikely that this regulation is likely to influence long-term investment decisions of pharmaceutical companies, especially as the results of the benefit assessment and the price negotiations depend on many other factors.