Statistical services

Analysis of clinical trial data in accordance with IQWiG and EU-HTA methodology
In many cases, Health Technology Assessment (HTA) procedures require statistical analyses that go beyond those included in the clinical trial report. These include, for example, the calculation of effect measures such as Hedges’ g, responder analyses using specified cut-off values, or additional subgroup analyses.
Our statisticians will handle the preparation and analyses of your trial data for you – professionally and in accordance with the methodology of the German Institute for Health Technology Assessment (IQWiG) and the European HTA Coordination Group.
Meta-analyses
Meta-analyses based on randomized controlled trials (RCTs) represent the gold standard of evidence-based medicine with evidence level 1a. Within the framework of benefit assessment procedures, they are routinely required for the synthesis of clinical data. We can support you in the systematic preparation and selection of studies and carry out the calculation of the meta-analyses on your behalf.
Indirect comparisons
Indirect comparisons are always necessary in benefit assessments when no direct comparative studies are available for a new active substance against the appropriate comparator therapy determined by the authorities. We identify studies suitable for an indirect comparison on your behalf and carry out the indirect comparison for you.
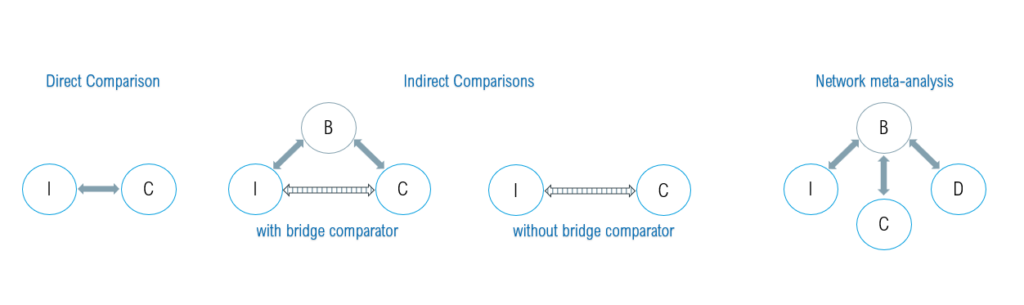
Figure 1: Various methods for comparing clinical evidence: direct treatment comparisons, indirect treatment comparisons with and without a bridge comparator, and network meta-analyses. B = bridge comparator; C = comparator; I = intervention; D= other treatment (for illustrative purposes)
Indirect treatment comparison using a bridge comparator
When two or more randomized controlled trials are available and include a common comparator (e.g. Trial 1: I vs. B and Trial 2: C vs. B), it can be used as an anchor point or “bridge comparator to compare the treatment effects of treatments A and B. We can support you in preparing and conducting indirect treatment comparisons or network meta-analyses.
Indirect treatment comparisons without a bridge comparator
Indirect treatment comparisons of two or more treatment groups without a bridge comparator present major challenges, as it cannot be assumed per se that the treatment groups are structurally equivalent. This necessitates rigorous adjustment for potential confounding factors (so-called confounders) that could cause bias for the treatment outcome, and is required by HTA authorities. We can carry out a comprehensive confounder search and the statistical analyses required for the indirect comparison on your behalf.
