The current draft bill on statutory health insurance contribution rate stabilization (BStabG) sets out the health policy objective of tighter expenditure control — with direct implications for pricing, market access, and reimbursement in the German market.
An overview of the key changes for innovative products:
- Discontinuation of the AMNOG guardrails and the combination rebate
Removal of complex and controversial legislations.
➡️ Price negotiations can once again be based more heavily on the extent of the additional benefit
➡️ The risk of a 20% discount on combination drugs no longer applies.
- Dynamic manufacturer discount (§ 130a):
Revenue-based budgeting for the pharmaceutical sector.
➡️ The costs of innovation in the pharmaceutical sector are capped and spread across all patented drugs.
➡️ The benefit assessment as a pricing tool is becoming less important.
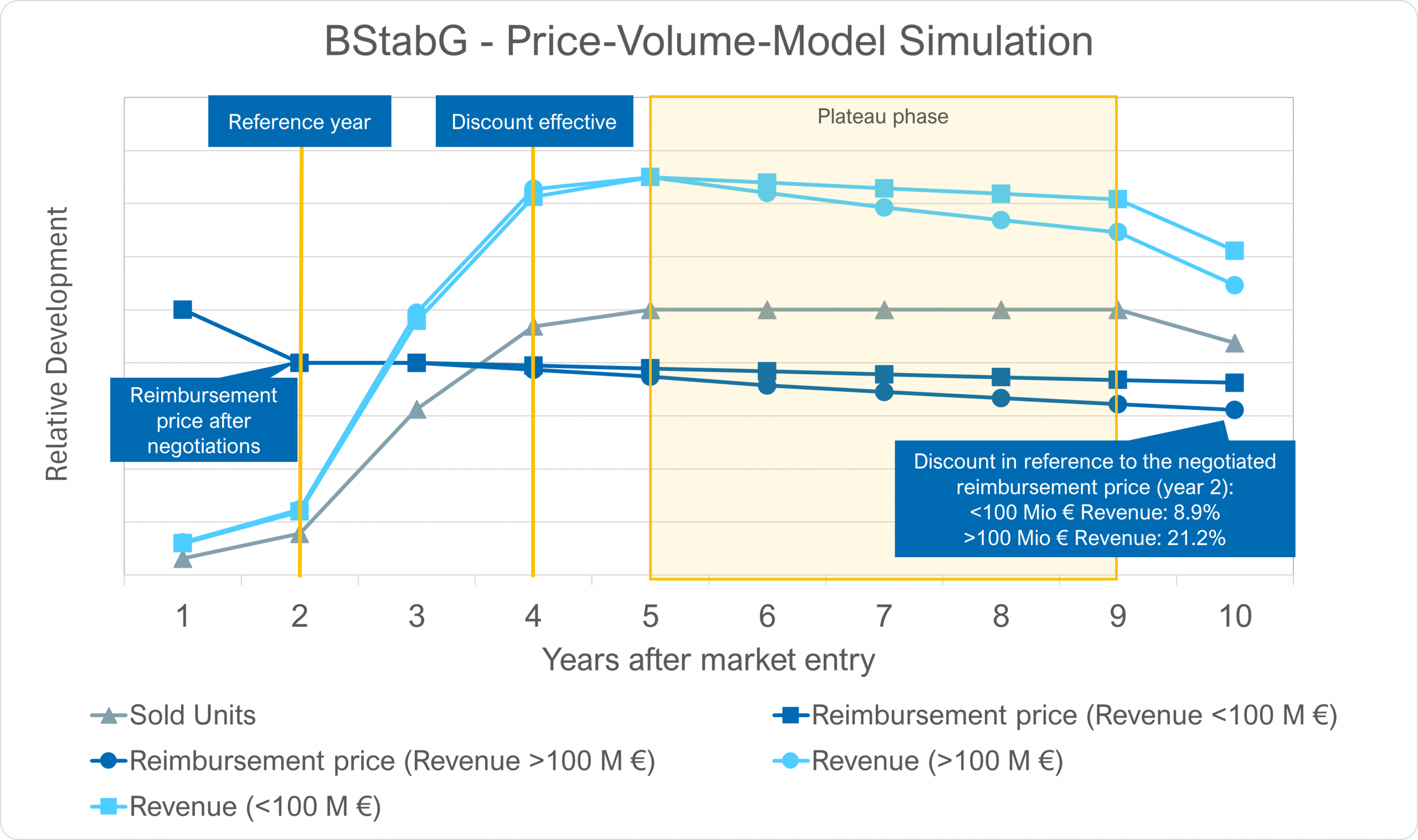
- Strengthening of the price-volume model as a central control instrument:
Growth and sales volume are systematically integrated into price development.
➡️ The arbitration board’s decision-making practice will be implemented as a legal requirement.
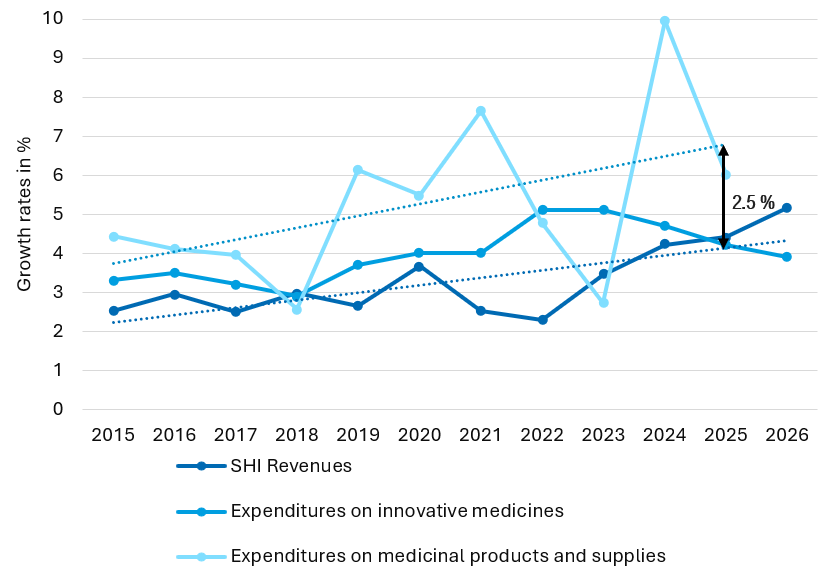
The central instrument for cost-control in the pharmaceutical market will be the dynamic manufacturer discount limiting the drug expenditure to the income of the statutory health insurances. The implicit drug budget will have impact on the pricing for new innovative drugs and will create interdependencies between prices for new drugs and patent-protected drugs that are already in the market.