Reimbursement of Medical Devices
- Strategic Consulting on the Reimbursement of Medical Devices
- Preparation of Annex V Applications
- NUB Applications

Market access and reimbursement of medical devices
The reimbursement of medical devices in Germany differs fundamentally from that of medicinal products: CE certification of a medical device does not automatically confer eligibility for reimbursement. This is subject to various conditions, depending on whether the medical device is used in an outpatient or inpatient setting, whether it involves a new method, and the type of medical device. For example, medical methods associated with medical devices in the outpatient sector are subject to what is known as ‘reservation of approval’, meaning they are only eligible for reimbursement once the G-BA has given the method a positive assessment. In contrast, the ‘reservation of prohibition’ principle applies in the inpatient sector: methods may be used until the G-BA excludes them from reimbursement following a negative assessment.
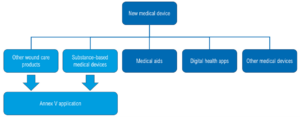
Figure 1: Overview of different categories of medical devices
Our Services
Our expertise lies in preparing Annex V applications for other wound care products and substance-based medical devices, as well as in compiling the dossier for the Joint Clinical Assessment (JCA).
Preparation of Annex V applications
Appendix V of the Medicines Directive (AM-RL) lists medical devices that have received a positive assessment and are therefore eligible for prescription at the expense of statutory health insurance (GKV). This regulation covers so-called ‘other wound care products’ and ‘substance-based medical devices.’ Inclusion in this positive list can be requested through an Annex V application. To do so, the medical device manufacturer must demonstrate the product’s appropriateness, medical necessity, and therapeutic benefit in the relevant patient population through studies of the highest possible level of evidence — ideally randomized controlled trials (RCTs). Once included in Annex V, the medical device is reimbursable only for the treatment of the patient population that was the subject of the evaluation.
Wound care products
Wound care products include dressings and other wound care products. While dressings primarily function by covering, absorbing, stabilizing, immobilizing, or compressing the wound, other wound care products have therapeutic effects that go beyond these properties. They can thus actively influence wound healing in the human body through pharmacological, immunological, or metabolic mechanisms. Dressings are generally eligible for reimbursement immediately. Other wound treatment products, however, must be included in Annex V of the AM-RL and thus undergo an evaluation by the G-BA to become eligible for reimbursement. An exception applies to products that were already eligible for reimbursement prior to December 2nd, 2020 — these remain eligible for reimbursement under a statutory transitional provision that has been extended multiple times, currently until December 31st, 2026.
Examples: Antimicrobial wound care products (e.g., silver-containing, honey-containing), hydrogels, or hydrocolloid dressings
Substance-based medical devices
Substance-medical medical devices are substances or compositions that resemble medicinal products in their presentation and form of administration but, in contrast, primarily exert a physical rather than a pharmacological, immunological, or metabolic effect in the human body. They are also not immediately eligible for reimbursement. For these products as well, the therapeutic benefit must first be demonstrated through studies providing the highest possible level of evidence in an Annex V application.
Examples: Synthetic tears, certain irrigation solutions used during surgical procedures, carrier solutions for the administration of inhalants
Preparation of Dossiers for the JCA
Under Regulation (EU) 2021/2282, medical devices of risk classes IIb and III, as well as in vitro diagnostic medical devices, have been eligible for joint clinical assessment (JCA) since January 12th, 2025. In contrast to the phased implementation of JCAs for medicinal products at launch, the medical devices in question are selected by the Commission after obtaining a recommendation from the Coordination Group. The criteria for this focus primarily on unmet medical needs, potential impacts on patients, public health, or healthcare systems, and whether the product is the first in a new product category. Consequently, only selected medical devices will initially undergo a JCA process. This results in planning uncertainty, as it is not determined until late in the process whether a specific product must undergo the JCA process. Upon request, the medical device manufacturer must submit a dossier with the highest possible level of evidence and potentially comparisons across many PICOs.
Other categories of medical devices
In addition to the above, there are also the following types of medical devices, among others:
Medical aids
Medical aids are products that support medical treatment, compensate for disabilities, or prevent impending disabilities. They are characterized by the fact that they are generally used by patients at home. The medical aids eligible for reimbursement are listed in the Medical Aids Directory of the GKV-Spitzenverband.
Examples: crutches, prosthetics, respiratory aids, wheelchairs
Digital health apps (DiGAs)
DiGAs are app- or web-based medical devices that are prescribed by healthcare professionals. They are therefore also referred to as ‘apps on prescription’. The Federal Institute for Drugs and Medical Devices (BfArM) evaluates their clinical benefits and maintains a registry of DiGAs that can be prescribed.
Examples: Apps for treating back pain, anxiety disorders, migraines, or obesity
Other medical devices
In addition, there is a wide variety of medical devices whose reimbursement essentially depends on whether they are used in outpatient or inpatient settings. In the outpatient setting, medical devices that are part of a new examination or treatment method must undergo a method evaluation by the G-BA in accordance with section 135 of the German Social Code, Book V (SGB V) in order to become eligible for reimbursement (reservation of permission). Subsequently, in the event of a positive evaluation, a billing code is created in the German Uniform Assessment Standard (EBM). In the inpatient setting, medical devices are generally reimbursed within the framework of the case-based fixed sum system of diagnosis related groups (DRGs) but may be prohibited by the G-BA following a method evaluation according to section 137c of the SGB V (reservation of prohibition).
Examples: in vitro diagnostic devices (laboratory tests), diagnostic imaging equipment (e.g., MRI scanners), or products for surgical use (e.g., cardiac catheters)
