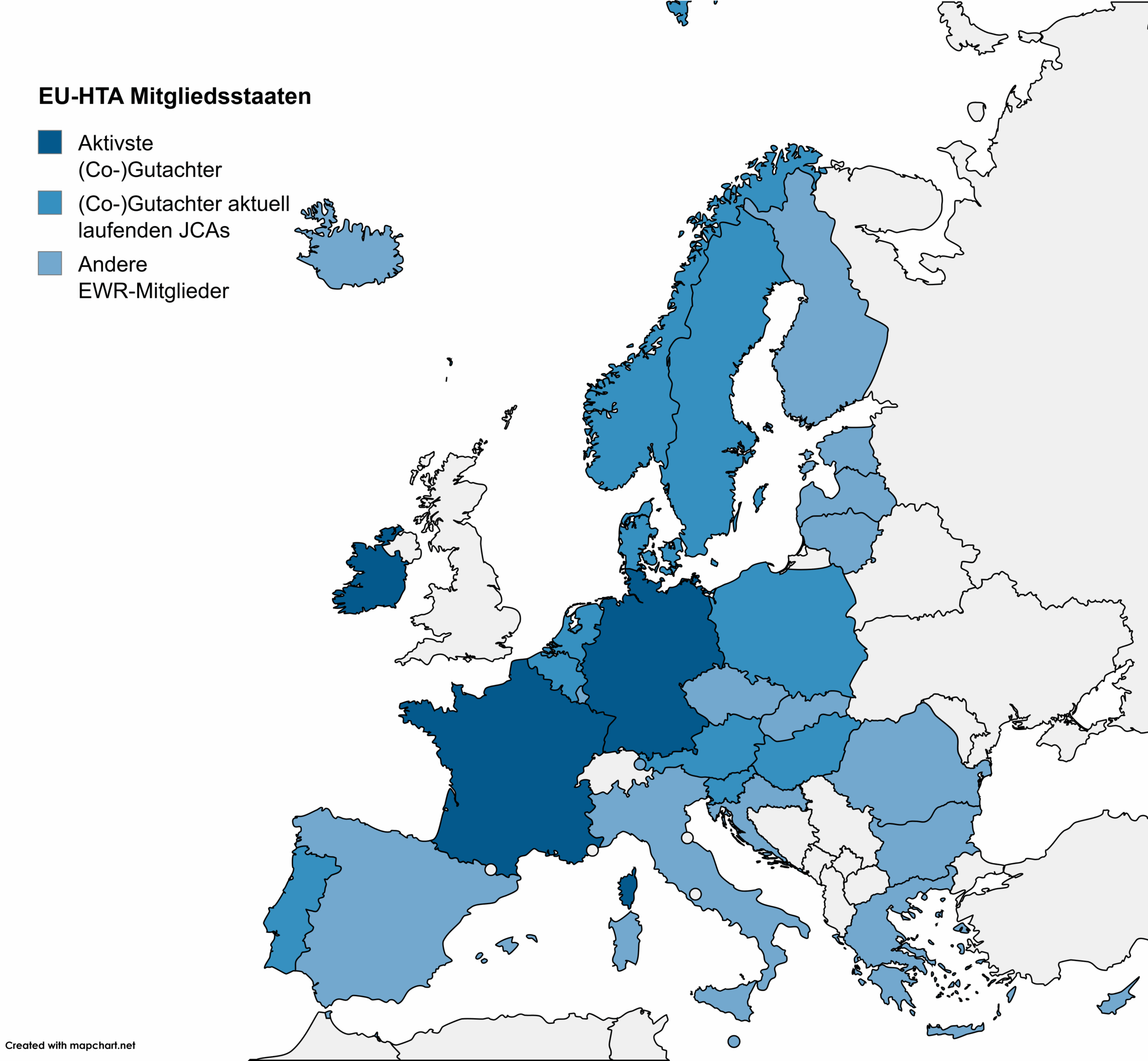
This month, the European Council adopted the regulation on the EU-wide assessment of health technologies (EU-HTA). At the same time, the first call for applications is now open for scientific advice within the framework of EUnetHTA.
In the open call of the EUnetHTA 21 JSC Secretariat, pharmaceutical companies can apply for parallel consultations with the EUnetHTA 21 and the European Medicines Agency (EMA). The aim of the consultations is to provide non-binding, scientific advice regarding the design of clinical trials to generate high-quality data for future benefit assessments. The EUnetHTA 21 Joint Consortium will be led by the National Health Care Institute of the Netherlands (ZIN). The Joint Federal Committee (G-BA) in Germany acts as the secretariat of the joint scientific consultations. Applications can be submitted to the G-BA email address EUnetHTA21-JSC@g-ba.de until December 7, 2021.