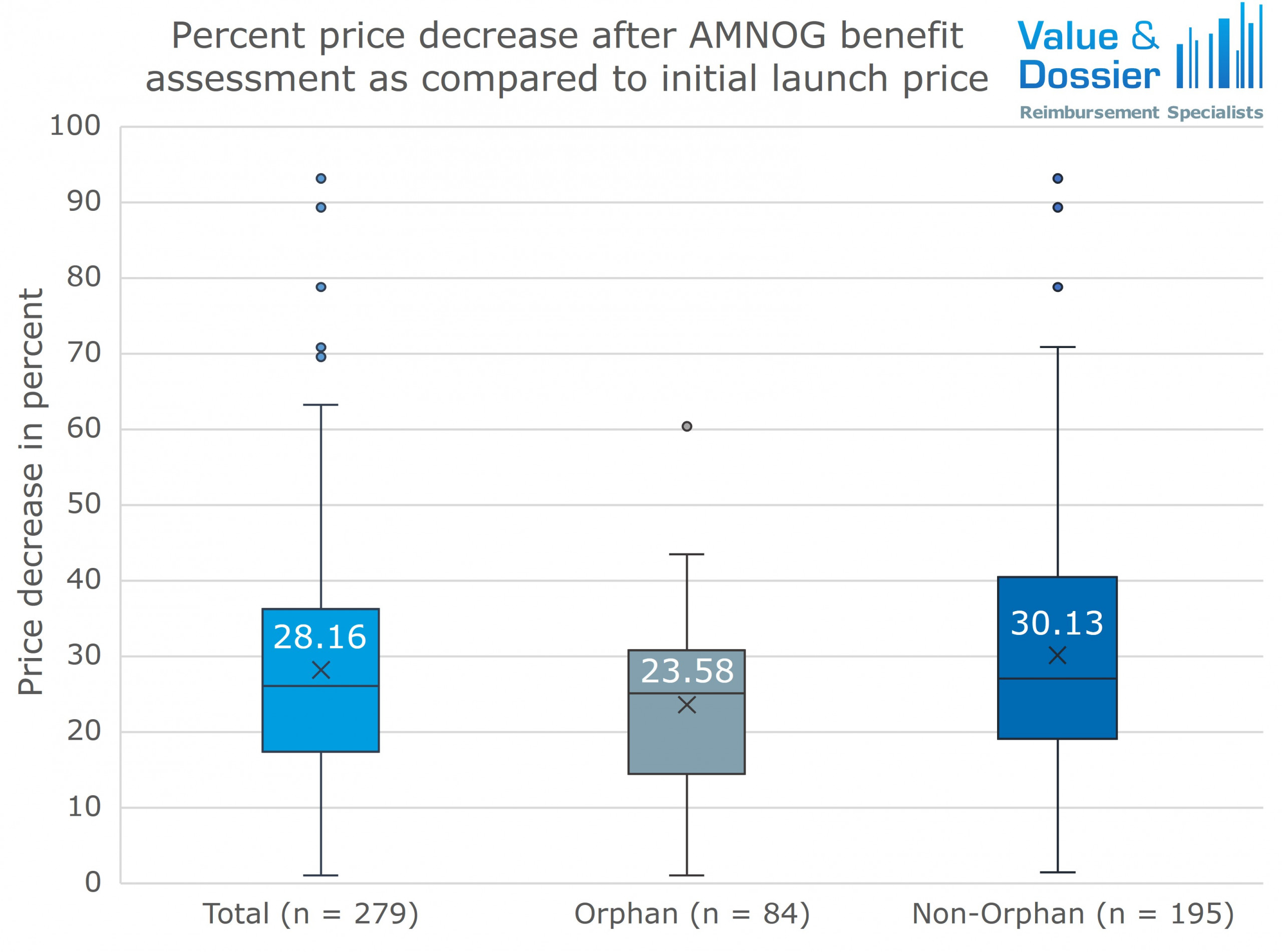
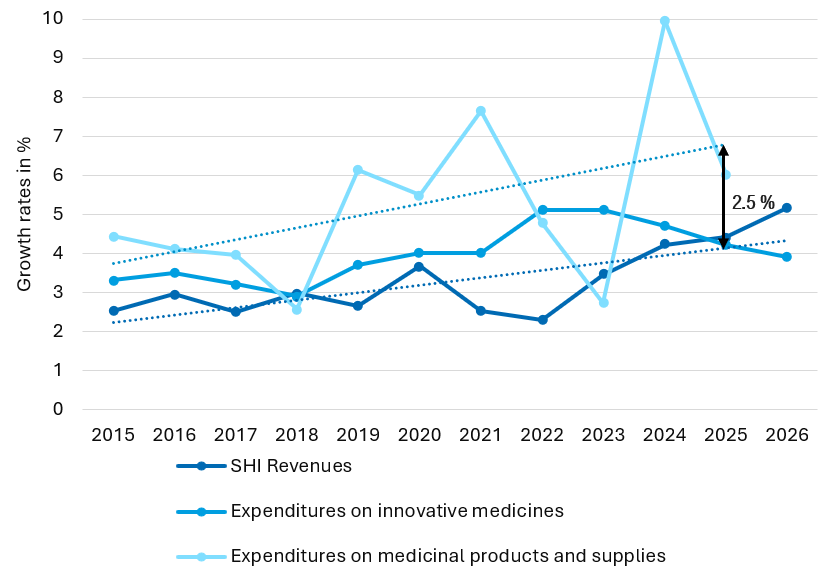
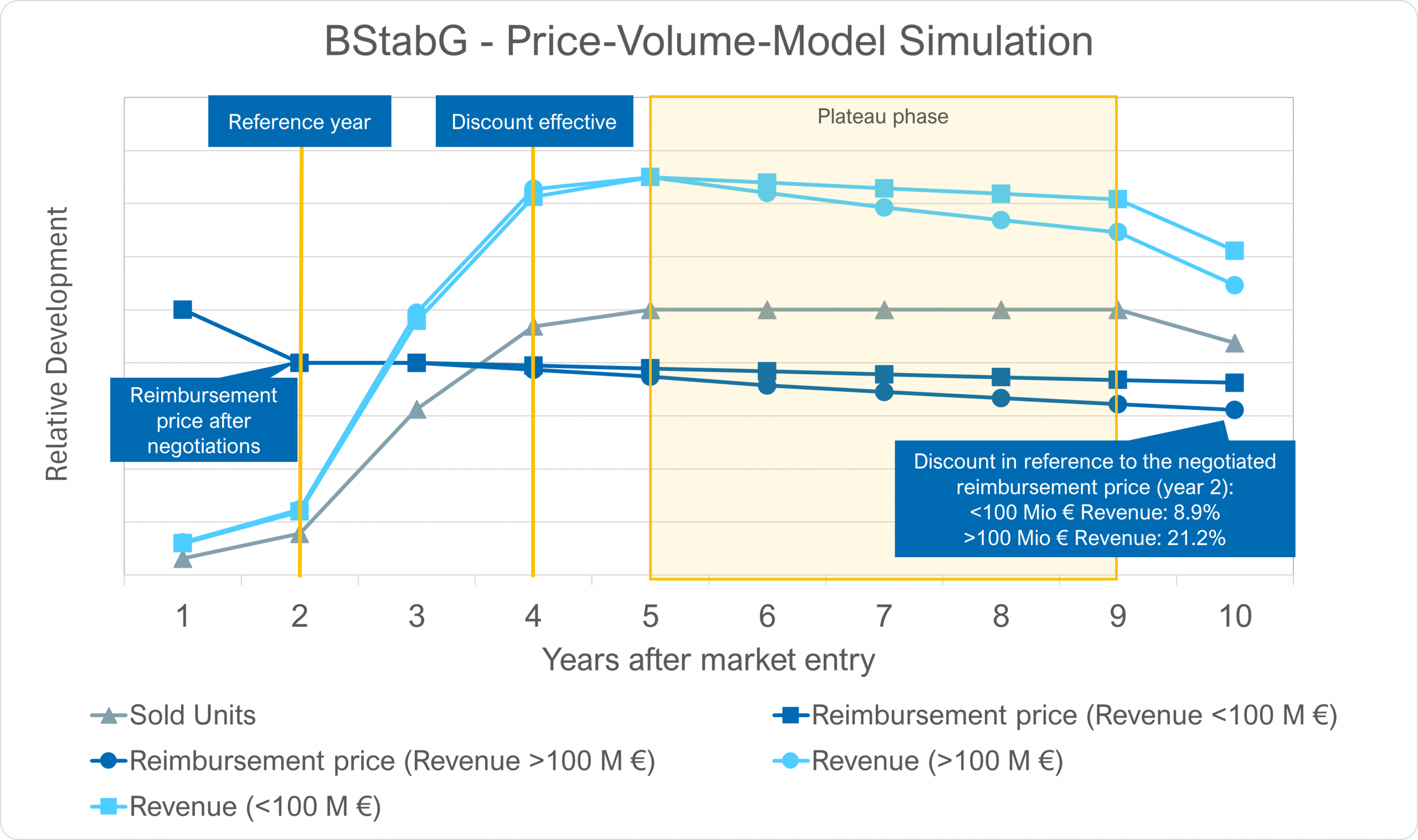
Is the new dynamic mandatory rebate proposed in the BStabG a sustainable solution for controlling pharmaceutical expenditure?
The dynamic manufacturer rebate is a regulatory misstep The planned introduction of a dynamic manufacturer rebate for patent-protected medicines (§ 130a para. 1b SGB V) as part of the BStabG (statutory health insurance contribution rate stabilization bill) raises significant regulatory questions — p…