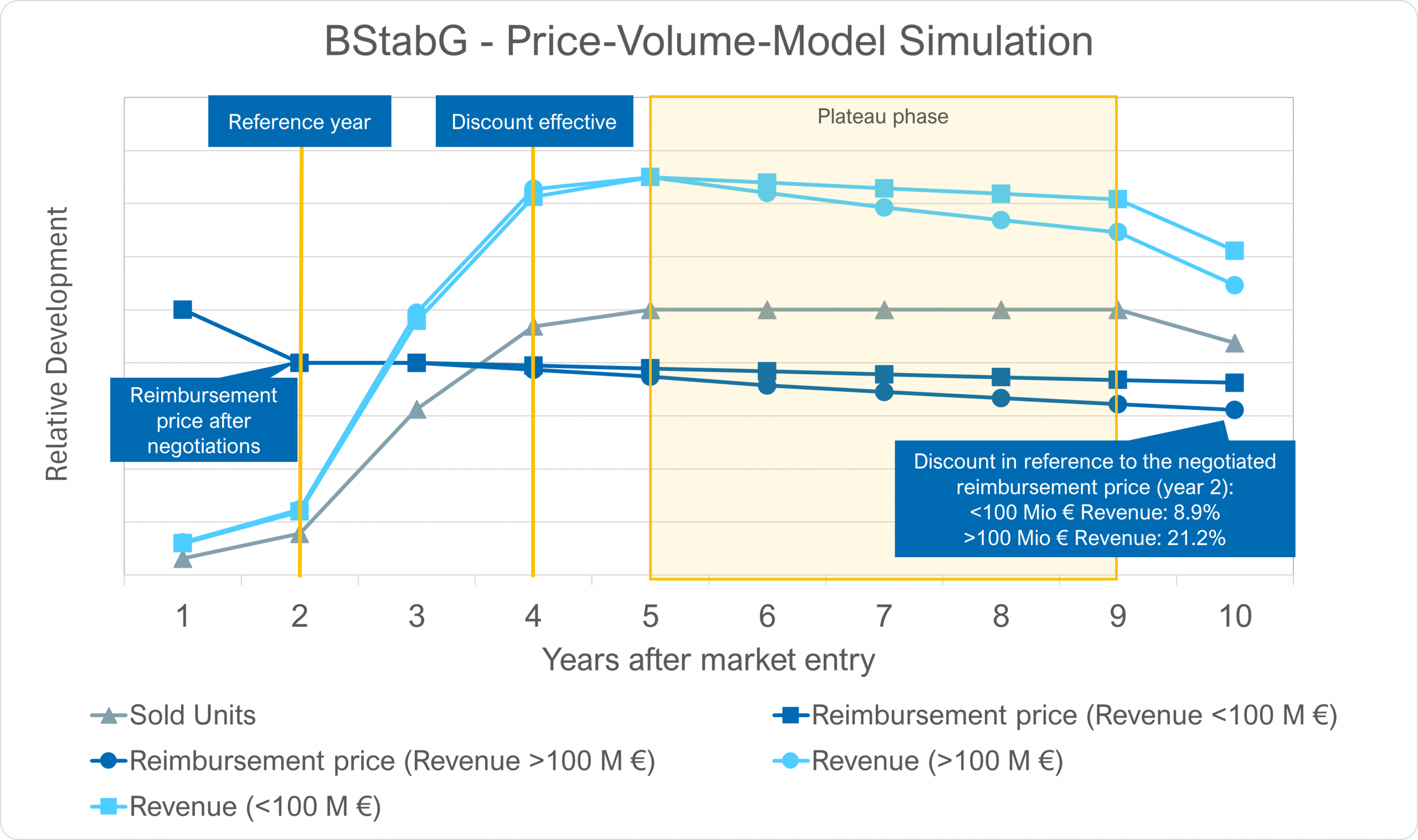
Deep Dive: Price-Volume Model (§130b)
The price-volume regulation shall no longer be subject to arbitration. The default mechanism kicks in if no agreement between the pharmaceutical company and the statutory health insurances is reached and adjusts the reimbursement amount annually (starting in the third calendar year after launch) usi…